Cancer Xenograft Models in Lung Cancer Research
Xenograft models are essential tools in lung cancer research, enabling the study of human tumor biology within a living organism. By implanting human lung cancer cells into immunodeficient mice, researchers can observe tumor progression, metastasis, and response to therapeutic agents in an environment that closely mimics human disease. These models provide critical insight into drug efficacy and mechanisms of action that cannot be fully captured through in vitro studies alone.
Lung cancers are heterogeneous diseases, with subtypes such as non-small cell lung carcinoma (NSCLC) and small cell lung carcinoma (SCLC) differing in their biology, genetic mutations, and treatment responses. Xenograft models enable precise evaluation of these diverse tumor types, allowing the development and testing of targeted therapies, immunotherapies, and combination regimens in a controlled and reproducible setting.
Understanding Lung Cancer
Lung cancer remains one of the leading causes of cancer-related mortality worldwide. NSCLC comprises the majority of cases and includes adenocarcinoma, squamous cell carcinoma, and large cell carcinoma, each with distinct histopathological and molecular characteristics. Common genetic alterations include mutations in EGFR, KRAS, ALK rearrangements, and MET amplifications, which influence tumor behavior and therapeutic susceptibility.
Small cell lung carcinoma, although less common, is highly aggressive and characterized by rapid growth and early metastasis. The distinct biology of SCLC requires specialized models to study therapeutic vulnerabilities and resistance mechanisms.
Role of Xenograft Models in Translational Lung Cancer Research
Lung cancer xenograft models serve as a critical bridge between laboratory research and clinical application. These in vivo systems allow for the testing of experimental drugs under physiological conditions, providing insight into tumor growth inhibition, metastasis, and toxicity. Importantly, xenografts replicate the tumor microenvironment, including interactions with stromal cells, blood vessels, and immune components, which impact therapeutic outcomes.
By employing xenograft models, researchers can evaluate the efficacy of novel targeted agents such as tyrosine kinase inhibitors, monoclonal antibodies, and immune checkpoint inhibitors. These models also facilitate the study of resistance mechanisms and the identification of predictive biomarkers. Altogen Labs offers comprehensive lung cancer xenograft services using a variety of well-characterized cell lines, delivering customizable protocols and detailed analytical support to accelerate drug development and translational research.
A549 Xenograft Model
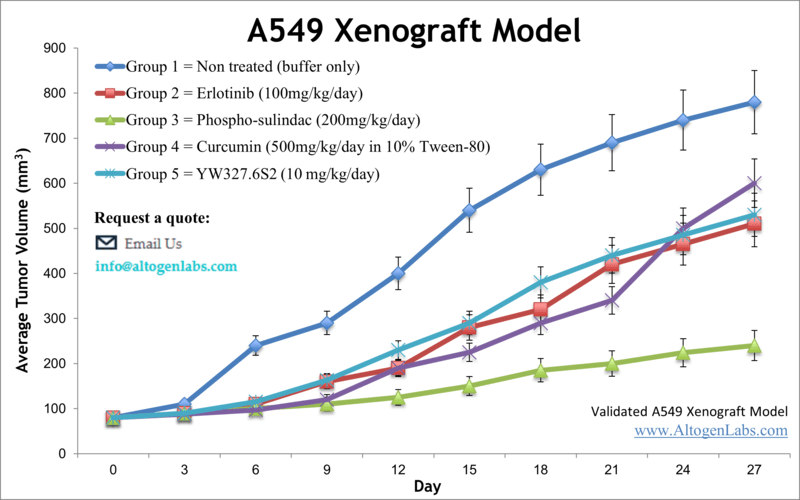
Overview of the A549 Xenograft Model
The A549 xenograft model is a widely accepted in vivo system for studying non-small cell lung cancer (NSCLC), particularly lung adenocarcinoma. A549 cells are derived from a 58-year-old Caucasian male with alveolar basal epithelial carcinoma and retain key characteristics of type II pneumocytes. This model is extensively used for evaluating tumor growth inhibition, angiogenesis, and response to chemotherapeutic agents and targeted therapies. Due to its consistent tumorigenicity and histological resemblance to human lung cancer, the A549 xenograft model remains a cornerstone in translational oncology research.
Scientific Background: A549 Cell Line
A549 cells display epithelial morphology and are characterized by wild-type KRAS and mutated TP53 genotypes. They are proficient in surfactant protein production and demonstrate robust responses to epidermal growth factor receptor (EGFR) signaling and DNA-damaging agents. These cells are non-squamous in origin, which makes them highly representative of lung adenocarcinoma—a subtype accounting for the majority of NSCLC cases. A549 cells are also widely used in studies related to oxidative stress, drug transport, epithelial barrier function, and gene regulation in the lung microenvironment. Their consistent in vitro behavior and ability to generate solid tumors in vivo make them highly valuable for lung cancer modeling.
Xenograft Study Protocol
Altogen Labs establishes A549 xenografts using immunodeficient athymic nude or NOD/SCID mice, typically aged 6–8 weeks. A549 cells are maintained under standardized conditions, verified for >98% viability, and prepared in serum-free media with or without Matrigel. Approximately 1×10⁶ to 5×10⁶ cells are injected subcutaneously into the hind flank. Tumor nodules are generally visible within 7–10 days and reach measurable size by day 10–14 post-injection.
Mice are randomized into treatment cohorts once tumors reach 100–150 mm³. Compound administration is performed via custom protocols tailored to dosing schedules, delivery routes (e.g., intraperitoneal, oral gavage, intravenous), and compound class. Tumor dimensions and body weight are recorded two to three times per week. Studies typically conclude within 3–5 weeks depending on tumor growth kinetics and observed therapeutic effects. All procedures follow IACUC-approved protocols in GLP-compliant facilities.
Endpoint Analysis and Data Output
Altogen Labs provides a comprehensive data package with every xenograft study, covering the full experimental timeline. Core deliverables include tumor growth curves, body weight trends, raw measurement data, dose-response comparisons, and high-resolution documentation of tumor sites. Optional downstream analysis includes necropsy, tissue preservation, histological staining, immunohistochemistry, protein and nucleic acid isolation, qPCR, Western blotting, and transcriptome profiling. Clients may define customized endpoint readouts such as apoptotic index, angiogenesis markers, or gene-specific expression changes. All services are designed for maximum reproducibility, scientific accuracy, and compliance with regulatory expectations.
Applications and Use Cases
The A549 xenograft model is highly applicable to a wide range of preclinical research areas, including drug screening, mechanism-of-action studies, and pharmacodynamics profiling in lung cancer. It is especially useful for evaluating EGFR pathway modulators, KRAS-targeted strategies, and DNA repair inhibitors. This model supports both monotherapy and combination therapy designs, allowing researchers to explore synergistic interactions between standard-of-care drugs and novel experimental compounds. Given its non-small cell lung cancer origin and extensive biological characterization, the A549 xenograft system is an ideal platform for advancing new therapeutic approaches in respiratory oncology.
Request an Instant Quote: https://altogenlabs.com/request-quote/a549-xenograft-model-services/
Learn more: A549 Xenograft Model
H460 Xenograft Model
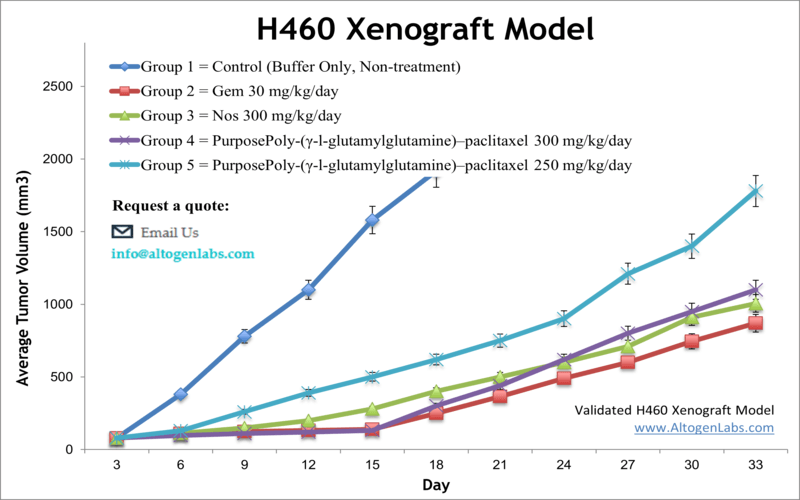
Overview of the H460 Xenograft Model
The H460 xenograft model is a well-characterized preclinical platform for studying large cell lung carcinoma, a subtype of non-small cell lung cancer (NSCLC). The NCI-H460 cell line was originally derived from a pleural effusion of a male patient with stage III NSCLC and is widely used in oncology research due to its high tumorigenicity, rapid growth kinetics, and well-documented genetic profile. As a subcutaneous xenograft, H460 tumors form reliably in immunodeficient mice and are used extensively for evaluating chemotherapeutic efficacy, molecular pathway inhibitors, and radiation sensitizers. This model is especially suited for studies investigating tumor hypoxia, apoptosis, and metabolic reprogramming in lung cancer.
Scientific Background: NCI-H460 Cell Line
The NCI-H460 cell line is classified as large cell carcinoma, a poorly differentiated subtype of NSCLC. It harbors mutations in TP53 and exhibits wild-type KRAS, making it an appropriate model for investigating pathways that drive proliferation, survival, and genomic instability. H460 cells grow as adherent monolayers in vitro and possess a highly glycolytic phenotype, characteristics that correlate with aggressive tumor behavior in vivo. Importantly, this cell line is sensitive to agents targeting mitochondrial respiration, DNA synthesis, and PI3K/AKT signaling, making it a versatile model for both traditional cytotoxic drug screening and targeted therapeutic development.
H460 cells are also commonly used in radiation biology due to their responsiveness to ionizing radiation and their utility in studying DNA damage repair mechanisms. These features make the model valuable in research that bridges lung cancer therapeutics, radiotherapy optimization, and biomarker validation.
Xenograft Study Protocol
Altogen Labs generates H460 xenografts using athymic nude or NOD/SCID mice aged 6–8 weeks. Cell preparations are assessed for viability exceeding 98% and suspended in sterile saline or Matrigel depending on the study design. Typically, 5×10⁶ cells in 100–150 μL are injected subcutaneously into the right or bilateral flanks. Tumors become palpable within 5–7 days and achieve measurable volumes by days 10–12.
When tumors reach 100–200 mm³, mice are randomized into treatment cohorts for compound dosing. Custom study protocols allow for precise control over administration route (oral, IV, IP, etc.), frequency, and dosing regimens. Tumor volume and animal health parameters are monitored multiple times per week. Studies generally run 3–5 weeks, depending on treatment objectives and tumor progression. All experiments are conducted under GLP-compliant conditions and IACUC oversight to ensure ethical and scientific integrity.
Endpoint Analysis and Data Output
Each H460 xenograft study includes a full dataset documenting tumor growth, treatment response, and health metrics. Standard deliverables include digital tumor measurements, growth inhibition curves, endpoint tumor weights, and photographic records. Optional downstream analyses include histology, TUNEL staining, immunohistochemistry, Western blotting, ELISA, RNA extraction, and gene expression profiling. Clients may also request pharmacodynamic marker evaluation or tissue bioavailability studies depending on study goals. All results are delivered in a publication-ready format and tailored to the client’s scientific and regulatory requirements.
Applications and Use Cases
The H460 xenograft model is widely used in lung cancer research, especially for evaluating anticancer drugs that target DNA replication, metabolic adaptation, or stress response pathways. It is particularly valuable for testing radiosensitizers, chemotherapy resistance mechanisms, and immunomodulatory agents in a tumor setting that mimics aggressive NSCLC behavior. Due to its fast and consistent tumor growth, the model supports rapid proof-of-concept studies, dose-response profiling, and combination therapy investigations. It is also frequently employed in PK/PD studies to understand drug behavior in tumor tissue.
Request an Instant Quote: https://altogenlabs.com/request-quote/h460-xenograft-model-services/
Learn more: H460 Xenograft Model
NCI-H226 Xenograft Model
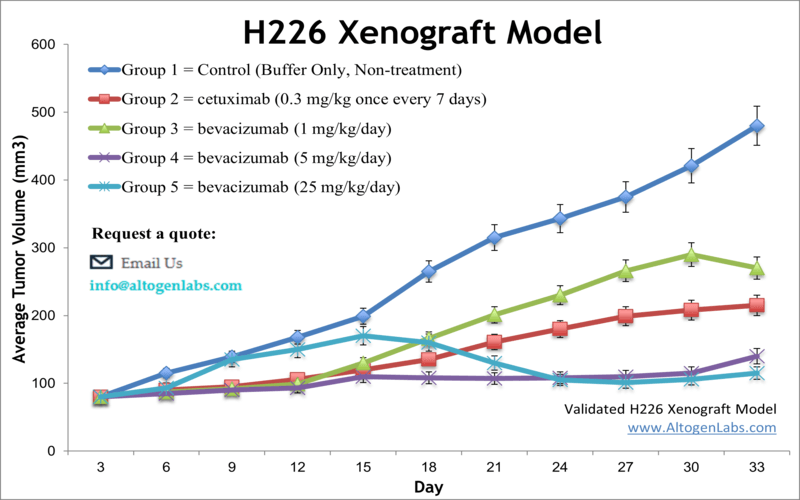
Overview of the NCI-H226 Xenograft Model
The NCI-H226 xenograft model is a valuable in vivo platform for studying squamous cell carcinoma (SCC) of the lung, a histological subtype of non-small cell lung cancer (NSCLC) that remains difficult to treat and poorly represented in preclinical models. Derived from a lung SCC tumor, the NCI-H226 cell line forms reproducible subcutaneous tumors in immunodeficient mice, making it a highly relevant tool for investigating tumor biology, chemotherapeutic response, and molecular signaling pathways specific to squamous lung cancers. The model is widely used for evaluating EGFR pathway inhibitors, radiation sensitizers, and agents targeting cell cycle progression and DNA repair.
Scientific Background: NCI-H226 Cell Line
NCI-H226 is a human lung squamous carcinoma cell line originally derived from a pleural effusion of a male NSCLC patient. Unlike many other NSCLC models that represent adenocarcinoma, H226 retains the morphological and genetic features of SCC, including high levels of cytokeratin expression, p63 positivity, and low surfactant protein production. The cell line harbors TP53 mutations and exhibits an epithelial phenotype, which is useful for dissecting pathways associated with cell adhesion, differentiation, and squamous cell lineage-specific vulnerabilities.
H226 cells have been used extensively to study mechanisms of cisplatin sensitivity, oxidative stress response, and EGFR-independent oncogenesis. Their tumorigenic behavior in vivo, combined with the capacity for molecular profiling and drug response assays, makes them a preferred model for research aimed at developing targeted treatments for SCC of the lung.
Xenograft Study Protocol
Altogen Labs conducts NCI-H226 xenograft studies in NOD/SCID or athymic nude mice aged 6–10 weeks. The cells are cultured to logarithmic growth phase, verified for >98% viability, and suspended in sterile PBS or Matrigel. Typically, 5×10⁶ cells in 100–150 μL are injected subcutaneously into the flank region. Tumors are usually palpable within 7–10 days and grow at a moderate rate, reaching treatment-ready volumes (100–150 mm³) by days 12–16.
Once tumors are established, mice are randomized into control and treatment arms. Drug administration routes and dosing schedules are fully customized, and tumor measurements are taken multiple times weekly using digital calipers. Studies usually run for 3–5 weeks depending on compound efficacy and growth kinetics. All procedures follow GLP-compliant workflows and IACUC-approved protocols, ensuring quality and ethical animal handling throughout the study.
Endpoint Analysis and Data Output
Comprehensive data packages are provided for each xenograft project and include tumor growth curves, endpoint tumor volumes and weights, body weight tracking, and high-resolution imaging of tumor sites. Optional analyses include immunohistochemistry, RNA and protein extraction, qPCR, Western blotting, TUNEL assays, and histopathological evaluation. Researchers may define specific biomarkers or molecular readouts for pharmacodynamic assessments. Altogen Labs also offers custom reporting formats designed to meet regulatory or publication requirements, with all data validated for accuracy and reproducibility.
Applications and Use Cases
The NCI-H226 xenograft model is uniquely suited for squamous lung cancer research, enabling preclinical testing of novel agents that target pathways often overlooked in adenocarcinoma-centric studies. It is used for evaluating drug combinations, radiation-enhancing agents, apoptosis inducers, and inhibitors of DNA repair enzymes. The model is also relevant for studying tumor microenvironment interactions, squamous lineage-specific vulnerabilities, and resistance mechanisms to traditional chemotherapies such as platinum-based agents.
With its relevance to a histological subtype of NSCLC that has few effective targeted therapies, the NCI-H226 xenograft system plays a crucial role in translational oncology, helping bridge the gap between molecular discoveries and clinical application in SCC treatment.
Request an Instant Quote: https://altogenlabs.com/request-quote/nci-h226-xenograft-model-services/
Learn more: NCI-H226 Xenograft Model
NCI-H1975 Xenograft Model
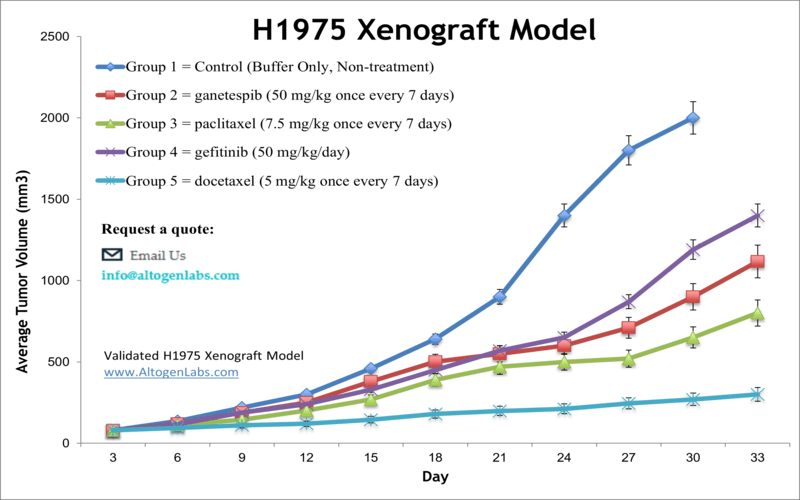
Overview of the NCI-H1975 Xenograft Model
The NCI-H1975 xenograft model is an advanced in vivo platform for studying non-small cell lung cancer (NSCLC) driven by EGFR mutations, specifically the L858R activating mutation and the T790M resistance mutation. These two mutations make the H1975 cell line a powerful tool for modeling acquired resistance to first- and second-generation EGFR inhibitors. When implanted subcutaneously in immunodeficient mice, H1975 cells form aggressive tumors suitable for testing EGFR-targeted therapies, resistance-overcoming compounds, and novel combinatorial strategies. This model is widely used in translational oncology for evaluating small molecule inhibitors, monoclonal antibodies, and genetic therapies directed at EGFR signaling and downstream pathways.
Scientific Background: NCI-H1975 Cell Line
The NCI-H1975 cell line was derived from a human lung adenocarcinoma and is known for harboring both the L858R point mutation in exon 21 and the T790M gatekeeper mutation in exon 20 of the EGFR gene. This combination reflects clinical cases of acquired resistance to first-generation tyrosine kinase inhibitors (TKIs) such as erlotinib and gefitinib. The cell line displays epithelial morphology and is highly dependent on EGFR signaling for growth and survival, making it a clinically relevant model for developing third-generation TKIs like osimertinib and other next-generation agents.
NCI-H1975 cells are widely utilized in preclinical studies focusing on drug resistance, pathway reactivation, and apoptotic evasion. Their reproducible growth, mutational specificity, and well-defined pharmacological responses make them indispensable for drug development programs targeting resistant lung adenocarcinomas.
Xenograft Study Protocol
Altogen Labs establishes NCI-H1975 xenografts in NOD/SCID or athymic nude mice, typically aged 6–8 weeks. Cells are cultured to exponential growth phase, tested for viability (>98%), and resuspended in sterile PBS or Matrigel prior to subcutaneous injection into the flank. A typical inoculum ranges from 3×10⁶ to 5×10⁶ cells in 100–150 μL. Tumor take is highly consistent, with nodules becoming visible by day 7–10 and reaching treatment size (100–200 mm³) within two weeks.
Once tumors reach the desired volume, mice are randomized into dosing groups. Altogen Labs provides fully customizable study designs, including route of administration (oral, IP, IV), dose levels, treatment frequency, and duration. Tumor growth and animal health metrics are collected two to three times weekly. The study duration typically ranges from 3 to 5 weeks, with early endpoints available for rapid assessment of efficacy. All procedures are performed in GLP-compliant facilities under IACUC-approved animal protocols.
Endpoint Analysis and Data Output
A standard NCI-H1975 xenograft study includes detailed tumor growth curves, endpoint tumor volumes and weights, and body weight tracking. Clients also receive high-resolution tumor imaging and raw data files suitable for publication or regulatory submission. Optional downstream services include pharmacokinetics/pharmacodynamics analysis, Western blotting, RNA and protein isolation, immunohistochemistry, TUNEL assay, qPCR, and next-generation sequencing. Customized reporting formats are available to support IND submissions, journal publications, or internal review processes. All data are validated for consistency, scientific rigor, and traceability.
Applications and Use Cases
The NCI-H1975 xenograft model is a gold standard for testing novel EGFR inhibitors, especially compounds targeting the T790M mutation or overcoming EGFR-related resistance. It is also highly relevant for evaluating the combination of TKIs with agents targeting downstream signaling pathways such as PI3K/AKT/mTOR or RAS/RAF/MEK. This model supports research into clonal evolution under drug pressure, mechanisms of resistance reactivation, and synthetic lethality strategies. Additionally, it provides a preclinical testing ground for therapies under development for use in patients with EGFR-mutant lung adenocarcinoma who have progressed on earlier-generation inhibitors.
Request an Instant Quote: https://altogenlabs.com/request-quote/nci-h1975-xenograft-model-services/
Learn more: NCI-H1975 Xenograft Model
DMS273 Xenograft Model
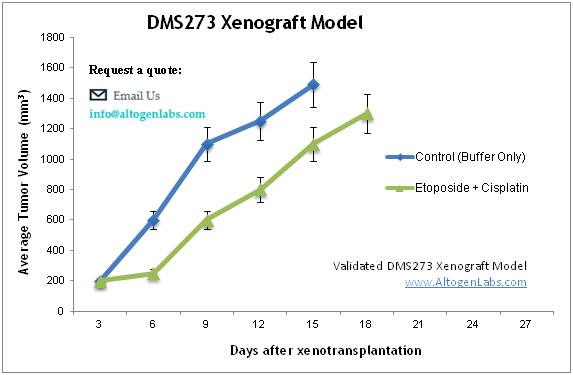
Overview of the DMS273 Xenograft Model
The DMS273 xenograft model is a clinically relevant in vivo system for studying small cell lung cancer (SCLC), a highly aggressive neuroendocrine tumor type characterized by rapid growth, early metastasis, and initial responsiveness to chemotherapy. Derived from a metastatic SCLC lesion, DMS273 cells reliably form subcutaneous tumors in immunodeficient mice, providing a high-fidelity platform for investigating therapeutic efficacy, treatment resistance, and tumor biology unique to SCLC. This model is particularly valuable for evaluating new chemotherapeutic agents, DNA damage response inhibitors, and experimental immuno-oncology combinations in a disease setting that remains largely underserved by targeted therapies.
Scientific Background: DMS273 Cell Line
The DMS273 cell line was established from the bone marrow metastasis of a Japanese male patient with diagnosed small cell lung carcinoma. Unlike some SCLC models, DMS273 cells grow adherently and express both epithelial and neuroendocrine markers, reflecting a classic SCLC phenotype. They are positive for neuron-specific enolase (NSE) and synaptophysin, and exhibit high proliferative capacity and susceptibility to DNA-damaging agents. The line carries mutations commonly associated with SCLC pathogenesis, including alterations in TP53 and RB1, which mirror genetic landscapes observed in clinical cases.
DMS273 cells have been widely used to model tumor progression, drug-induced apoptosis, and the molecular mechanisms underlying chemoresistance. Their robust tumorigenicity in vivo and neuroendocrine lineage specificity make them ideal for exploring novel treatment strategies in the context of high-grade pulmonary malignancies.
Xenograft Study Protocol
Altogen Labs conducts DMS273 xenograft studies using athymic nude or NOD/SCID mice aged 6–10 weeks. Cells are expanded under controlled culture conditions and verified for >98% viability prior to implantation. For tumor induction, 3×10⁶ to 5×10⁶ cells are injected subcutaneously in 100–150 μL of sterile media or Matrigel into the hind flank. Tumors typically become palpable within 5–8 days and grow rapidly, reaching treatment thresholds (100–200 mm³) within 10–14 days.
Once tumors are established, mice are randomized into treatment groups. Altogen Labs customizes every aspect of the study protocol, including compound formulation, administration route (oral, IP, IV), dosing schedule, and monitoring frequency. Tumor size and body weight are measured multiple times per week, and study duration is typically 3–5 weeks depending on experimental endpoints and therapeutic response. All animal work is conducted under IACUC-approved protocols in GLP-compliant facilities to ensure high standards of reproducibility and animal welfare.
Endpoint Analysis and Data Output
Clients receive a full study report that includes tumor growth curves, final tumor weights, body weight measurements, and photographic documentation of tumor sites. Additional services offered include histological evaluation, immunohistochemistry, TUNEL staining, Western blotting, RNA and protein extraction, and quantitative PCR. Studies can be tailored to include pharmacokinetics/pharmacodynamics (PK/PD) analysis, toxicity assessment, and molecular biomarker discovery. Altogen Labs ensures data integrity and offers custom formatting of results for regulatory filings, academic publishing, or internal drug development programs.
Applications and Use Cases
The DMS273 xenograft model is a highly relevant tool for preclinical evaluation of anticancer agents targeting small cell lung carcinoma. It is particularly suited for testing compounds that induce apoptosis, disrupt cell cycle progression, or impair DNA repair pathways in aggressive neuroendocrine tumors. The model supports evaluation of chemotherapy combinations, emerging immunotherapies, and resistance-modulating agents. Its rapid and reproducible growth makes it ideal for short-term efficacy studies, while its lineage specificity supports research into the biology and clinical treatment of high-grade pulmonary cancers that currently lack effective targeted options.
Request an Instant Quote: https://altogenlabs.com/request-quote/
Learn more: DMS273 Xenograft Model
LL2 Allograft Model
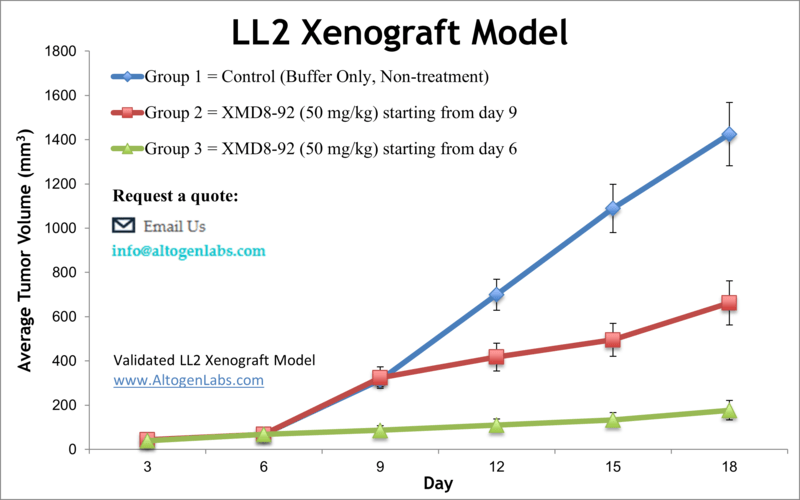
Overview of the LL2 Allograft Model
The LL2 allograft model (Lewis Lung Carcinoma) is a syngeneic, immunocompetent mouse model widely used in lung cancer research. Unlike xenograft systems that rely on immunodeficient mice, the LL2 model allows for the study of tumor-immune interactions, immune checkpoint inhibition, and tumor microenvironment dynamics in fully functional C57BL/6 mice. Originally derived from a spontaneous lung carcinoma in this mouse strain, LL2 cells are highly tumorigenic and metastasis-prone, offering a fast, reliable, and immunologically relevant system for evaluating novel anticancer agents and immunotherapies.
Scientific Background: LL2 Cell Line
Lewis Lung Carcinoma (LL2 or LLC1) is a murine lung tumor cell line derived from a C57BL/6 mouse. The cells grow rapidly in vitro and in vivo, display an epithelial-like morphology, and are known for their ability to form solid tumors with high metastatic potential—particularly to the lungs when injected intravenously or subcutaneously. LL2 tumors exhibit aggressive behavior, hypoxic cores, and a pro-inflammatory microenvironment, which makes them especially suitable for studying angiogenesis, tumor progression, and immune modulation.
The LL2 model is routinely used to evaluate immuno-oncology agents, including checkpoint inhibitors (such as PD-1 or CTLA-4 blockade), vaccine candidates, and cytokine therapies. Due to the syngeneic nature of the system, it supports intact T-cell, B-cell, macrophage, and dendritic cell responses—providing a translational bridge between molecular oncology and immune response research.
Allograft Study Protocol
Altogen Labs establishes LL2 tumors in female C57BL/6 mice, typically aged 6–8 weeks. Cells are cultured to logarithmic growth phase, tested for >98% viability, and injected subcutaneously into the flank at a concentration of 1×10⁶ to 5×10⁶ cells in 100 μL sterile PBS or Matrigel. Tumors become visible within 4–6 days and grow rapidly, reaching 100–200 mm³ within the first 10–12 days post-inoculation.
Once tumors reach the desired size, mice are randomized into treatment groups, and therapeutic dosing is initiated. Study designs are fully customizable and can include monotherapies, combination regimens, or immune checkpoint blockade. Drug administration routes include intraperitoneal, intravenous, and oral delivery. Tumor volumes and body weights are recorded multiple times per week. Study duration typically ranges from 2 to 4 weeks, depending on therapeutic efficacy and tumor kinetics. All studies are performed under GLP-compliant conditions and IACUC-approved protocols to ensure reproducibility and ethical animal care.
Endpoint Analysis and Data Output
The LL2 allograft service includes complete data documentation, from tumor onset to final endpoint. Deliverables include tumor growth curves, digital caliper measurements, tumor weights, body weights, and photographic records. Optional services include histopathological analysis, immunohistochemistry for immune infiltration (CD8⁺ T cells, macrophages, Tregs), RNA and protein isolation, ELISA for cytokine levels, qPCR, and flow cytometry to evaluate immune cell populations. Clients may also request PK/PD studies or immune profiling to assess treatment-specific effects.
Applications and Use Cases
The LL2 allograft model is highly applicable for evaluating novel immunotherapies and small molecule inhibitors in lung cancer within an intact immune system. It is frequently used to study checkpoint blockade, tumor vaccination strategies, angiogenesis inhibitors, and T-cell–modulating agents. Additionally, it provides a robust framework for understanding tumor-host interactions, resistance mechanisms, and combinatorial treatment efficacy in immuno-oncology. Its compatibility with genetically engineered mouse models and ease of use make it a key tool in preclinical lung cancer research.
Request an Instant Quote: https://altogenlabs.com/request-quote/ll2-xenograft-model-services/
Learn more: LL2 Allograft Model
Calu-3 Xenograft Model
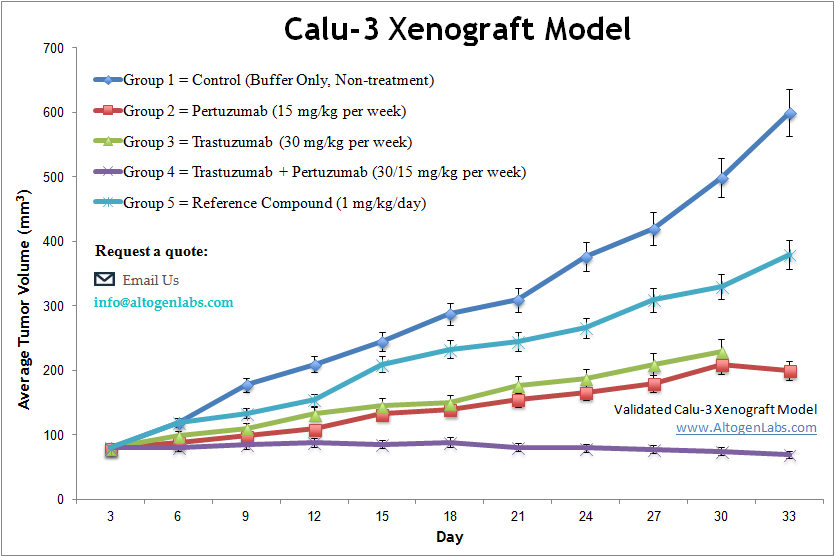
Overview of the Calu-3 Xenograft Model
The Calu-3 xenograft model is a well-established in vivo system for studying human lung adenocarcinoma with epithelial characteristics. Derived from a metastatic lung tumor, Calu-3 cells form well-differentiated, slowly growing tumors in immunodeficient mice, making this model especially suitable for long-term therapeutic studies and investigations into airway epithelium biology, drug absorption, and mucosal transport. The Calu-3 xenograft model supports research into both standard chemotherapeutic compounds and emerging targeted therapies aimed at epithelial lung cancers.
Scientific Background: Calu-3 Cell Line
The Calu-3 cell line was derived from a lung adenocarcinoma resected from the pleural effusion of a 25-year-old male patient. Unlike many lung cancer cell lines, Calu-3 cells exhibit a polarized epithelial morphology and are capable of forming tight junctions, producing mucins, and expressing high levels of E-cadherin. These traits make them a favored model for simulating human airway barrier function in addition to their relevance in lung cancer biology.
Calu-3 cells are known to express wild-type KRAS and moderate levels of EGFR, making them suitable for investigating mechanisms of cell adhesion, barrier disruption, and epithelial-to-mesenchymal transition (EMT). Their high transepithelial electrical resistance (TEER) in vitro and well-structured tumor histology in vivo further enhance their value as a model for drug delivery research and tumor epithelial integrity under therapeutic pressure.
Xenograft Study Protocol
Altogen Labs establishes Calu-3 xenografts in athymic nude or NOD/SCID mice aged 6–10 weeks. The cells are expanded under sterile conditions, tested for >98% viability, and injected subcutaneously at a concentration of 5×10⁶ to 10×10⁶ cells in 100–150 μL of Matrigel or PBS. Calu-3 tumors grow at a slower pace compared to more aggressive lung carcinoma lines, typically becoming palpable by days 10–14 and reaching treatment-appropriate volumes (100–200 mm³) within 2–3 weeks.
Once tumors are established, mice are randomized into treatment and control groups. Dosing regimens, delivery routes, and frequency are tailored to the experimental goals, with tumor volume and animal weight tracked multiple times weekly. Studies usually run 4–6 weeks to accommodate the model’s growth kinetics. All procedures are conducted under GLP-compliant conditions and IACUC-approved protocols to ensure ethical and reproducible research practices.
Endpoint Analysis and Data Output
Each Calu-3 xenograft study includes a comprehensive data package that documents tumor progression, therapeutic response, and animal health. Deliverables include tumor volume curves, endpoint tumor weights, body weight logs, and high-resolution images of tumor sites. Additional analytical services include histopathological evaluation, immunohistochemistry (e.g., for tight junction proteins, EGFR, Ki-67), RNA and protein extraction, Western blotting, ELISA, and qPCR. Studies can also be customized to evaluate changes in epithelial structure, mucin production, or EMT-associated markers.
Applications and Use Cases
The Calu-3 xenograft model is ideal for preclinical research involving well-differentiated lung adenocarcinomas and epithelial cell-targeted therapies. It supports drug efficacy studies, tumor biology investigations, and airway-targeted drug delivery research. Its unique epithelial features make it particularly relevant for evaluating agents that affect cellular polarity, barrier integrity, and mucosal interactions. Additionally, it serves as a bridge between oncology and pulmonary drug development, offering insights into how epithelial cancers respond to treatment while maintaining physiological features similar to human airway tissue.
Request an Instant Quote: https://altogenlabs.com/request-quote/calu-3-xenograft-model-services/
Learn more: Calu-3 Xenograft Model
Calu-6 Xenograft Model
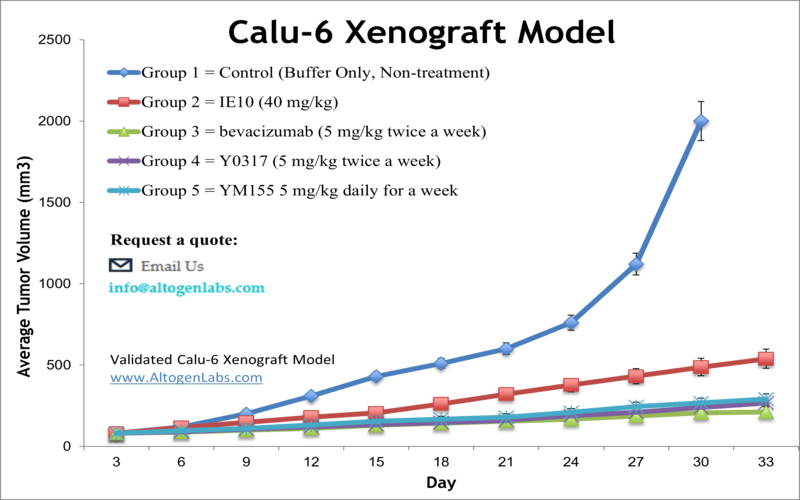
Overview of the Calu-6 Xenograft Model
The Calu-6 xenograft model is a widely used in vivo platform for studying undifferentiated human lung adenocarcinoma. Derived from a poorly differentiated tumor, Calu-6 cells form fast-growing subcutaneous tumors in immunodeficient mice, making this model suitable for time-sensitive drug efficacy testing, chemoresistance studies, and evaluation of novel therapeutics in non-small cell lung cancer (NSCLC). Its aggressive growth characteristics and reproducible tumor formation make it an ideal model for preclinical screening of cytotoxic agents and molecular pathway inhibitors.
Scientific Background: Calu-6 Cell Line
The Calu-6 cell line was established from a human lung adenocarcinoma and is characterized by a fibroblast-like morphology and a lack of tight junctions, in contrast to more differentiated lines such as Calu-3. These cells exhibit mesenchymal features and are negative for mucin production, E-cadherin expression, and tight junction proteins—indicating a dedifferentiated phenotype with high plasticity. This makes Calu-6 especially useful for studying epithelial-to-mesenchymal transition (EMT), tumor invasiveness, and metastasis.
Calu-6 cells carry mutations relevant to NSCLC biology, including p53 inactivation, and exhibit intermediate expression of growth factor receptors such as EGFR. Their rapid proliferation rate and responsiveness to DNA-damaging agents or anti-mitotic compounds further contribute to their utility in cancer drug development.
Xenograft Study Protocol
Altogen Labs establishes Calu-6 xenografts using immunodeficient nude or NOD/SCID mice aged 6–10 weeks. Cells are cultured to log-phase growth and tested for >98% viability prior to injection. For tumor induction, 5×10⁶ to 10×10⁶ cells are resuspended in 100–150 μL of Matrigel or PBS and injected subcutaneously into the flank. Tumors typically become palpable within 5–7 days and grow quickly, reaching treatment volumes (100–200 mm³) within 10–14 days.
Once tumors are established, mice are randomized into groups for compound dosing. Altogen Labs offers full customization of the experimental design, including dosing schedule, route of administration (oral, IP, or IV), and treatment duration. Tumor volumes and body weights are recorded multiple times per week. Studies generally last 3–4 weeks depending on tumor kinetics and therapeutic objectives. All procedures are GLP-compliant and conducted under IACUC-approved protocols.
Endpoint Analysis and Data Output
Each Calu-6 xenograft study includes a comprehensive data report featuring tumor growth curves, endpoint tumor weights, body weight monitoring, and tumor imaging. Optional downstream analyses include histopathology, immunohistochemistry, Western blotting, RNA/protein extraction, and qPCR. Biomarker evaluation, pharmacokinetic/pharmacodynamic assessments, and toxicity screening can be integrated into the study upon request. All data outputs are validated for accuracy and reproducibility and are formatted to support regulatory filings or peer-reviewed publications.
Applications and Use Cases
The Calu-6 xenograft model is well-suited for research focused on poorly differentiated NSCLC, particularly in studies evaluating agents that target EMT, mesenchymal markers, and cell cycle regulation. It is commonly used for drug screening, resistance mechanism studies, and preclinical assessment of combination therapies. Due to its aggressive growth and undifferentiated phenotype, the model also provides insights into tumor progression and metastatic behavior, making it a versatile system for translational oncology research.
Request an Instant Quote: https://altogenlabs.com/request-quote/calu-6-xenograft-model-services/
Learn more: Calu-6 Xenograft Model
H1155 Xenograft Model
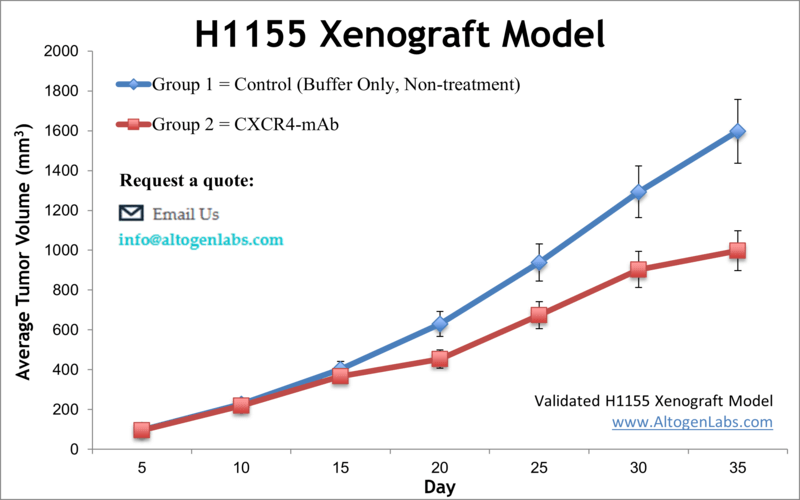
Overview of the H1155 Xenograft Model
The H1155 xenograft model is an established preclinical system used for studying small cell lung cancer (SCLC) in vivo. This model is derived from a human lung carcinoma that exhibits neuroendocrine features, high mitotic index, and rapid tumor growth, making it a reliable and aggressive platform for evaluating anticancer agents. H1155 xenografts are particularly useful for drug screening studies targeting DNA replication, cell cycle progression, and pro-apoptotic pathways. Their consistent tumor formation and fast-growing kinetics make them a preferred model for high-throughput efficacy testing and time-sensitive therapeutic evaluations.
Scientific Background: H1155 Cell Line
The NCI-H1155 cell line was established from a lung carcinoma of a male patient and is classified as a variant form of small cell lung carcinoma. These cells exhibit a neuroendocrine phenotype, including expression of markers such as synaptophysin and chromogranin A. H1155 cells have a high proliferation rate, grow in semi-adherent clusters in vitro, and display anchorage-independent growth characteristics, consistent with their tumorigenic nature.
Genetically, the H1155 line carries mutations commonly found in SCLC, including alterations in the p53 and RB1 tumor suppressor genes. These features contribute to unchecked cellular proliferation and resistance to apoptotic stimuli—hallmarks of aggressive lung cancer subtypes. This cell line is suitable for modeling both early-stage drug responses and acquired resistance mechanisms in neuroendocrine lung malignancies.
Xenograft Study Protocol
Altogen Labs establishes H1155 xenograft tumors using immunodeficient athymic nude or NOD/SCID mice, typically 6–8 weeks of age. The cells are cultured to exponential growth phase, tested for >98% viability, and injected subcutaneously into the flank region at concentrations of 5×10⁶ to 10×10⁶ cells in 100–150 μL of PBS or Matrigel. Tumors usually become palpable within 5–7 days and exhibit rapid growth, reaching treatment initiation volumes of 100–200 mm³ within 10–12 days post-injection.
After tumor establishment, animals are randomized into treatment groups. Altogen Labs customizes all dosing protocols, including compound formulation, administration route (IV, IP, or oral), and treatment frequency. Tumor growth is monitored with digital calipers, and animal health parameters such as weight and behavior are recorded multiple times per week. Studies generally last between 3–5 weeks depending on tumor kinetics and experimental endpoints, and all procedures are conducted in GLP-compliant, IACUC-regulated facilities.
Endpoint Analysis and Data Output
Each H1155 xenograft study includes a comprehensive data package that documents tumor volume progression, endpoint tumor measurements, body weight trends, and high-resolution tumor imaging. Optional endpoints include histopathology, immunohistochemistry for neuroendocrine markers, Western blotting, RNA/protein isolation, TUNEL assay, qPCR, and gene expression profiling. Pharmacokinetic and pharmacodynamic services can also be integrated into the protocol. All data are reviewed for scientific validity and formatted for use in regulatory submissions or peer-reviewed publication.
Applications and Use Cases
The H1155 xenograft model is valuable for preclinical evaluation of chemotherapeutic agents, DNA-damage response inhibitors, and novel drug combinations targeting neuroendocrine and small cell lung carcinomas. It supports studies into treatment-induced apoptosis, resistance development, and biomarkers associated with high-grade lung tumors. The model’s aggressive tumor kinetics and robust reproducibility make it ideal for assessing short-term drug effects and for validating molecular targets in rapidly proliferating lung cancer cells. Researchers investigating therapies for difficult-to-treat lung cancer variants benefit from the H1155 system’s close alignment with clinical features of SCLC and neuroendocrine tumors.
Request an Instant Quote: https://altogenlabs.com/request-quote/h1155-xenograft-model-services/
Learn more: H1155 Xenograft Model
H1703 Xenograft Model
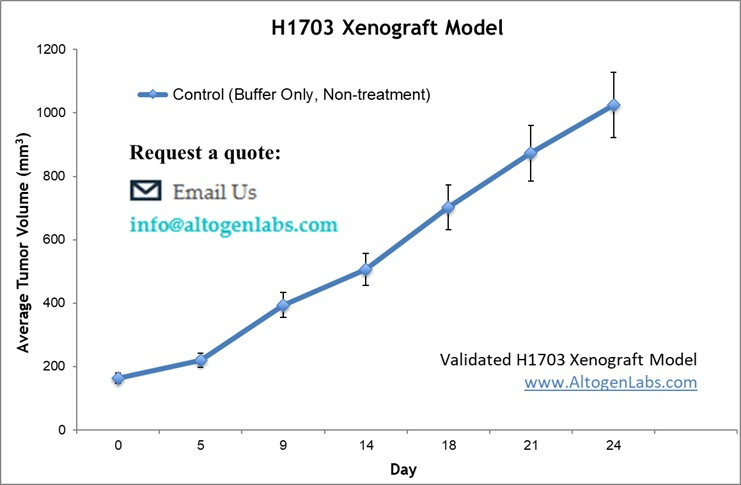
Overview of the H1703 Xenograft Model
The H1703 xenograft model is an in vivo platform used for studying human lung squamous cell carcinoma (SCC). Derived from a poorly differentiated squamous carcinoma of the lung, H1703 cells form rapidly growing tumors in immunodeficient mice, offering a robust and reproducible system for evaluating novel anticancer agents, targeted therapies, and combination regimens specific to squamous histology. This model is especially relevant for preclinical drug development efforts aiming to address therapeutic resistance and aggressive behavior observed in lung SCC.
Scientific Background: H1703 Cell Line
The H1703 cell line originates from a lung squamous cell carcinoma patient and is characterized by epithelial morphology typical of SCC. These cells express markers such as cytokeratins and p63, which are hallmarks of squamous differentiation. H1703 cells demonstrate a high proliferative rate and are known to carry genetic alterations common in lung SCC, including amplification of the FGFR1 gene and mutations affecting tumor suppressor pathways.
H1703 cells respond to therapies targeting cell cycle regulation, receptor tyrosine kinases, and apoptosis pathways, making them valuable for mechanistic studies and therapeutic screening. Their ability to form solid tumors in vivo with aggressive growth kinetics enables efficient assessment of anti-proliferative and cytotoxic agents in a preclinical context.
Xenograft Study Protocol
Altogen Labs establishes H1703 xenografts in immunodeficient nude or NOD/SCID mice aged 6–10 weeks. Cells are cultured under sterile conditions to log-phase growth and tested for >98% viability before implantation. For tumor induction, 5×10⁶ to 10×10⁶ cells are suspended in 100–150 μL of Matrigel or PBS and injected subcutaneously into the flank. Tumors typically become palpable within 7–10 days and grow rapidly, reaching volumes suitable for treatment initiation (100–200 mm³) within 12–15 days.
Following tumor establishment, animals are randomized into treatment groups, and dosing regimens are customized according to study requirements. Compound administration routes include oral, intravenous, and intraperitoneal delivery. Tumor volume and body weight measurements are recorded multiple times per week. Study durations typically span 3–5 weeks, depending on tumor growth kinetics and therapeutic response. All studies comply with GLP standards and are conducted under IACUC-approved protocols to ensure scientific rigor and animal welfare.
Endpoint Analysis and Data Output
Each H1703 xenograft study provides a detailed report that includes tumor growth curves, endpoint tumor weights, body weight monitoring, and photographic documentation of tumors. Additional analyses offered include histopathology, immunohistochemistry for squamous markers (e.g., cytokeratin 5/6, p63), Western blotting, RNA and protein extraction, and qPCR. Studies may be further customized to include pharmacokinetic/pharmacodynamic (PK/PD) analyses, toxicity profiling, and biomarker evaluation to support drug development pipelines and translational research.
Applications and Use Cases
The H1703 xenograft model is an essential tool for preclinical testing of novel therapies targeting lung squamous cell carcinoma. It supports investigations into drug resistance mechanisms, cell signaling pathways, and combination therapies aimed at overcoming aggressive tumor growth. The model is frequently used to evaluate receptor tyrosine kinase inhibitors, cell cycle modulators, and apoptosis-inducing agents. Its reproducible tumorigenicity and well-characterized molecular profile make it highly valuable for both academic research and pharmaceutical drug discovery focused on squamous lung cancer.
Request an Instant Quote: https://altogenlabs.com/request-quote/
Learn more: H1703 Xenograft Model
H1993 Xenograft Model
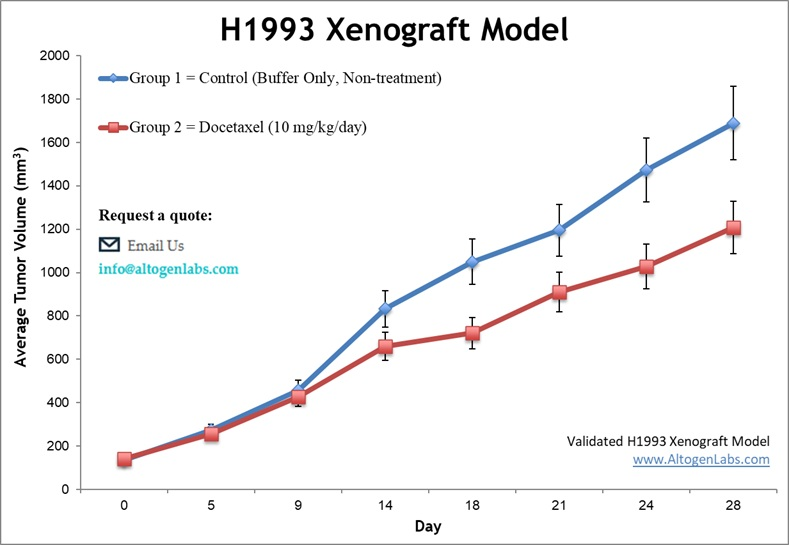
Overview of the H1993 Xenograft Model
The H1993 xenograft model is an established in vivo system for studying human lung adenocarcinoma, particularly tumors characterized by MET gene amplification. This model provides a valuable platform for investigating MET-driven oncogenesis and testing targeted therapies aimed at MET receptor tyrosine kinase. H1993 tumors grow reliably in immunodeficient mice, enabling robust evaluation of drug efficacy, resistance mechanisms, and combination regimens in a clinically relevant lung cancer subtype.
Scientific Background: H1993 Cell Line
The H1993 cell line was derived from a metastatic lung adenocarcinoma patient and is notable for its high-level amplification of the MET proto-oncogene. This genetic alteration leads to constitutive activation of MET signaling, promoting tumor cell proliferation, survival, invasion, and angiogenesis. H1993 cells exhibit epithelial morphology and express biomarkers typical of lung adenocarcinoma, including cytokeratins and EGFR.
Due to their MET dependency, H1993 cells serve as an ideal model for evaluating MET inhibitors, monoclonal antibodies, and small molecule kinase inhibitors. They are also used to study downstream signaling pathways such as PI3K/AKT and MAPK, which contribute to tumor growth and therapy resistance.
Xenograft Study Protocol
Altogen Labs establishes H1993 xenografts in athymic nude or NOD/SCID mice aged 6–10 weeks. Cells are cultured to logarithmic growth phase and tested for >98% viability before injection. Tumor initiation involves subcutaneous injection of 5×10⁶ to 10×10⁶ cells suspended in 100–150 μL of Matrigel or PBS into the flank region. Tumors become palpable within 7–10 days and typically reach treatment volumes (100–200 mm³) within 2–3 weeks.
After tumor establishment, mice are randomized into treatment groups for administration of investigational compounds. Dosing protocols, routes (oral, IP, IV), and schedules are tailored to experimental goals. Tumor volume and animal weights are monitored regularly. Study durations vary from 3 to 6 weeks, depending on tumor growth rates and therapeutic responses. All procedures comply with GLP standards and IACUC guidelines.
Endpoint Analysis and Data Output
Each H1993 xenograft study includes comprehensive data documentation with tumor growth curves, endpoint tumor weights, body weight records, and tumor imaging. Additional analytical services include histopathology, immunohistochemistry for MET and downstream effectors, RNA/protein extraction, Western blotting, qPCR, and biomarker profiling. Pharmacokinetic/pharmacodynamic assessments and toxicity analyses are available upon request. Data are validated for reproducibility and formatted for regulatory submissions or publication.
Applications and Use Cases
The H1993 xenograft model is instrumental for evaluating MET-targeted therapies in lung adenocarcinoma. It supports preclinical testing of kinase inhibitors, monoclonal antibodies, and combination regimens aimed at overcoming MET-driven tumor progression. Researchers also utilize this model to investigate mechanisms of acquired resistance and to identify predictive biomarkers. Its well-characterized genetic profile and consistent tumor growth make it a powerful tool for translational lung cancer research.
Request an Instant Quote: https://altogenlabs.com/request-quote/
Learn more: H1993 Xenograft Model
HCC827 Xenograft Model
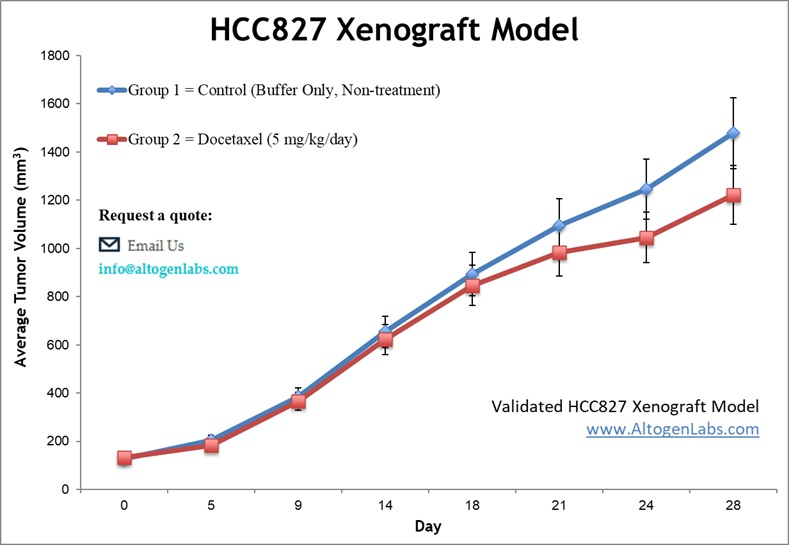
Overview of the HCC827 Xenograft Model
The HCC827 xenograft model is a widely used preclinical platform for studying lung adenocarcinoma characterized by activating mutations in the epidermal growth factor receptor (EGFR) gene. This model is particularly valuable for evaluating targeted therapies against EGFR, including tyrosine kinase inhibitors (TKIs). HCC827 tumors grow consistently in immunodeficient mice, providing a reliable system for investigating drug efficacy, resistance mechanisms, and combination treatments in non-small cell lung cancer (NSCLC).
Scientific Background: HCC827 Cell Line
The HCC827 cell line was derived from a lung adenocarcinoma patient and harbors an exon 19 deletion in the EGFR gene, resulting in constitutive activation of the receptor. This mutation drives uncontrolled cell proliferation and survival, making HCC827 an ideal model to study EGFR-dependent tumor biology and therapeutic response. These cells exhibit epithelial morphology and express lung-specific markers, maintaining key features of human lung adenocarcinoma.
HCC827 cells demonstrate high sensitivity to EGFR inhibitors such as erlotinib and gefitinib, but they can also develop resistance through secondary mutations or bypass signaling pathways. This makes the xenograft model particularly useful for testing new generations of EGFR-targeted agents and combination therapies aimed at overcoming resistance.
Xenograft Study Protocol
Altogen Labs establishes HCC827 xenografts in athymic nude or NOD/SCID mice aged 6–10 weeks. Cells are cultured to exponential growth phase and verified for >98% viability before implantation. For tumor induction, 5×10⁶ to 10×10⁶ cells are suspended in 100–150 μL of Matrigel or PBS and injected subcutaneously into the flank. Tumors become palpable within 7–10 days and typically reach treatment volumes (100–200 mm³) within 2–3 weeks.
After tumor establishment, mice are randomized into treatment groups with customized dosing schedules, routes of administration (oral, intravenous, or intraperitoneal), and treatment duration. Tumor volume and animal body weight are recorded multiple times per week. Studies generally last 3–6 weeks, depending on tumor growth kinetics and experimental objectives. All work is performed under GLP-compliant conditions and IACUC-approved protocols.
Endpoint Analysis and Data Output
Each HCC827 xenograft study includes detailed reports with tumor growth curves, endpoint tumor weights, body weight monitoring, and photographic documentation. Additional analyses include histopathology, immunohistochemistry for EGFR and downstream signaling molecules, Western blotting, RNA and protein extraction, qPCR, and biomarker profiling. Pharmacokinetic and pharmacodynamic analyses, as well as toxicity assessments, can be incorporated. Data outputs are validated and formatted to support regulatory submissions or scientific publications.
Applications and Use Cases
The HCC827 xenograft model is essential for preclinical evaluation of EGFR-targeted therapies in lung adenocarcinoma. It supports drug efficacy testing, studies of resistance mechanisms, and development of next-generation TKIs and combination regimens. This model is also valuable for biomarker discovery and pharmacodynamic assessments. Its genetic profile and reproducible tumor growth make it a cornerstone in translational research on EGFR-driven NSCLC.
Request an Instant Quote: https://altogenlabs.com/request-quote/
Learn more: HCC827 Xenograft Model
